Joseph Napoli
Ruth Okey Professor of Nutrition and Toxicology
Research Description
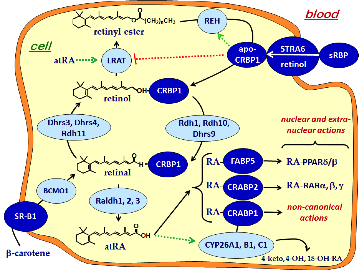
The Napoli lab studies regulation of the metabolon that generates the autocoid all-trans-retinoic acid (atRA) from retinol (vitamin A), and the function of atRA in in modulating intermediary metabolism (energy balance), diabesity, adipogenesis, bone health (osteoporosis) and blood glucose control. We use a combination of analytical chemistry, bioimaging, biochemistry and molecular and cell biology to determine the physiological effects of atRA.
Diabesity
We have individually knocked out two of the retinol dehydrogenases (Rdh) that catalyze the first reaction in the metabolon that converts retinol into atRA. The knockouts were created to determine the precise physiological function of endogenous atRA. Each Rdh catalyzes the same reaction, suggesting that each generates RA for distinct processes. Knockouts of Rdh1 and Rdh10 confirmed this hypothesis.
The Rdh1 knockout has a very precise and limited phenotype with major consequences—it becomes fatter than a wild-type mouse, despite not eating more and not decreasing activity. The null mouse, however, does not show the developmental defects caused by low atRA. In contrast to many models of adiposity, which require a high-fat diet to induce abnormal weight gain, the Rdh1-null mouse gains excess weight (37%) when fed a normal (low-fat) diet, and has a reduced core body temperature. This makes the Rdh1-null mouse a unique model to generate insight into gradual weight gain as humans age, and into weight gain related to regulating heat production, as opposed to energy expenditure. We have found that Rdh1 generates atRA to enhance lipid mobilization and fatty acid oxidation specifically in brown adipose tissue, explaining why its absence allows fat storage at the expense of maintaining body temperature.
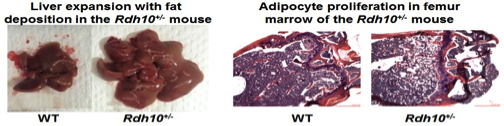
Most homozygous Rdh10-null mice pups die during gestation (embryo day 12.5) from severe forelimb, internal organs and forebrain abnormalities. Therefore, we prepared stem cells from heterozygotes and tested their ability to produce either adipocytes (fat-forming cells) or osteoblasts (bone-forming cells). Stem cells from the heterozygotes generated far more adipocytes and far fewer osteoblasts than stem cell from wild-type mice. This observation was confirmed in vivo. Lack of one allele of Rdh10 (Rdh10+/- mice) decreased tissue atRA modestly, but heightened sensitivity to diet-induced obesity, created glucose intolerance and insulin resistance, and induced non-alcoholic fatty liver disease (NAFLD) in males.
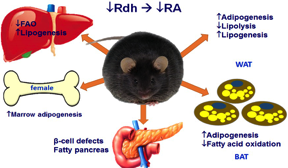
Females alone suffered adipocyte proliferation in bone (early onset osteoporosis). Rdh10+/- mice also suffer from pancreas and muscle degeneration. Low-dose atRA corrected these pathologies. The consequences of a modest decrease in tissue atRA suggest that impaired retinol activation may contribute to diabesity, and low-dose atRA therapy may ameliorate adiposity and its sequelae of glucose intolerance and insulin resistance. This model will be used to study the mechanisms whereby atRA directs stem cells to differentiate into adipocytes or osteoblasts, and to study the impact of atRA on NAFLD.
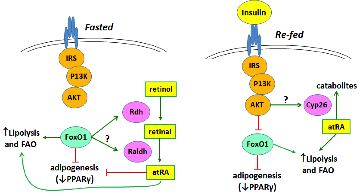
Insulin control of atRA biosynthesis
We have found that refeeding, glucose, and insulin decrease atRA concentrations in liver. This occurs through glucose stimulated insulin secretion suppressing Rdh1 and Rdh10 mRNA through arresting transcription and shortening their mRNA elimination half-lives. Insulin ejects the transcription factor FoxO1 from the nucleus, thereby preventing its transcription induction of Rdh expression. These results show that insulin and atRA exert counter balancing effects on regulating energy status. Ongoing work examines the precise mechanism of FoxO1 controlling Rdh transcription, and the effects of atRA on FoxO1 expression, and the physiological consequences of varying atRA concentrations with feeding and fasting.
The nervous system
The hippocampus serves as the center of memory formation and spatial navigation. Damage to the hippocampus prevents new memories from forming and causes spatial disorientation. For example, loss of short-term memory and disorientation noted in early onset Alzheimer’s disease reflects hippocampus damage. Likewise, low atRA or loss of atRA signaling also prevents spatial learning and memory, because the hippocampus requires atRA for neurogenesis and synaptogenesis. This lab was the first to show that atRA promotes synaptogenesis in the hippocampus by relieving translation suppression of an AMPA receptor subunit and CAMKIIα kinase, two proteins essential for synaptogenesis. Our ensuing studies were the first to show that a nuclear transcription factor regulated by atRA, RARa, also functions as a translation suppresser in RNA granules of hippocampus neurons. The lab continues to study the mechanisms of generating atRA in the hippocampus and its mechanisms of action (see also the connection to fetal alcohol spectrum disorder, below).
Fetal alcohol spectrum disorder (FASD)
The ability of ethanol to cause liver to mobilize its substantial stores of vitamin A has been known for many decades. Not understood have been the mechanisms of the mobilization, the disposition of the retinol, and most importantly, the biological consequences of ethanol’s interaction with retinoid signaling, although many studies have addressed these issues. We developed highly sensitive and specific analytical assays based on LC/MS/MS to resolve and quantify the geometric isomers of RA that occur in vivo, and HPLC/UV assays to quantify retinol and its storage form retinyl esters (RE), and used the assays to determine the impact of ethanol on the concentrations of retinoids in tissues. We confirmed that ethanol causes a massive decrease in RE and retinol in liver, but surprising found that ethanol prompts increases of atRA in testis, hippocampus and cortex—three tissues that require measured amounts of atRA for normal function. The increases seemed sufficient to contribute to the harmful effects of ethanol consumption, as 2-fold increases in atRA can be toxic. After dam ethanol consumption, increases in embryo hippocampus and cortex were as high as 50-fold. We are studying the impact of this increase on RA-regulated genes and processes and in post-natal behavior.
Education
- Ph.D. University of Michigan, Medicinal Chemistry
Publications
Altered vitamin A homeostasis and increased size and adiposity in the rdh1-null mouse. M. Zhang, P. Hu, C.R. Krois, M.A. Kane, J.L. Napoli. FASEB J. 21:2886-2896 (2007).
All-trans-retinoic acid stimulates translation and induces spine formation in hippocampal neurons through a membrane-associated RARα. N. Chen, J.L. Napoli. FASEB J. 22:236-245 (2008).
The nuclear transcription factor RARα associates with neuronal RNA granules and suppresses translation. N. Chen, B. Onisko, J.L. Napoli. J. Biol. Chem. 283:20841-20847 (2008).
Retinoic acid from the meninges regulates cortical neuron generation. J.A. Siegenthaler, A.M. Ashique, K. Zarbalis, K.P. Patterson, J.H. Hecht, M.A. Kane, A.E. Folias, Y. Choe, S.R. May, T. Kume, J.L. Napoli, A.S. Peterson, S.J. Pleasure. Cell 139:597-609 (2009).
Identification of 9-cis-retinoic acid as a pancreas-specific autacoid that attenuates glucose-stimulated insulin secretion. M.A. Kane, A.E. Folias, A. Pingitore, M. Perri, K. Obrochta, C.R. Krois, E. Cione, J.-Y. Ryu, J.L. Napoli. Proc. Natl. Acad. Sci. U.S.A., 107:21884-21889 (2010).
Ethanol elevates physiological all-trans-retinoic acid levels in select loci through altering retinoid metabolism in multiple loci: a potential mechanism of ethanol toxicity. M.A. Kane, A.E. Folias, C. Wang, J.L. Napoli. FASEB J. 24:823-832 (2010).
CrbpI modulates glucose homeostasis and pancreas 9-cis-retinoic acid concentrations. M.A. Kane, A.E. Folias, A. Pingitore, M. Perri, C.R. Krois, J-Y. Ryu, E. Cione, J.L Napoli. Mol. Cell. Biol. 16:3277-3285 (2011).
Multiple retinol and retinal dehydrogenases catalyze all-trans-retinoic acid biosynthesis in astrocytes. C. Wang, M.A. Kane, J.L. Napoli. J. Biol. Chem. 268:6542-6553 (2011).
Insulin regulates retinol dehydrogenase expression and all-trans-retinoic acid biosynthesis through FoxO1. K.M. Obrochta, C.R. Krois, B. Campos, and J.L. Napoli. J. Biol. Chem. 290: 7259- 7268 (2015).
Modest decreases in endogenous all-trans-retinoic acid produced by a mouse Rdh10 heterozygote provoke major abnormalities in adipogenesis and lipid metabolism.
D. Yang, M.G. Vuckovic, C.P. Smullin, M. Kim, C.P. Lo, E. Devericks, H.S.Yoo, M.Tintcheva Y. Deng, J.L. Napoli. Diabetes. 67:662-673 (2018).
Rdh1 suppresses adiposity by promoting brown adipose adaptation to fasting and re-feeding. C.R. Krois, M.G. Vuckovic, P. Huang, C. Zaversnik, C.D. Liu, C.E. Gibson, M.R. Wheeler, K.M. Obrochta, J.H., Min, C.B. Herber CB, A.C.l Thompson, I.D. Shah ID, S.P. Gordon, M.K. Hellerstein, J.L. Napoli. Cell. Mol. Life Sci. 76:2425-2447 (2019).
Post-natal all-trans-retinoic acid biosynthesis. J.L.Napoli. Methods Enzymol. 637:27-54 (2020).
Retinoic acid exerts sexually dimorphic effects on muscle energy metabolism and function. Y. Zhao, M. Vuckovic, H.S. Yoo, N. Fox, A. Rodriguez, K. McKessy, J.L. Napoli. J. Biol. Chem. 297:101101 (2021).